Mårtensson, "Core-Level Binding Energies in Metals," J. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. The electron configuration of the Cobalt can be represented as the Ar 3d7 4s2 in its most accurate and precise form. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). The data are adapted from references 1-3. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. All values of electron binding energies are given in eV. 1967, 47, 1300.Įlectron binding energies Electron binding energies for cobalt. 
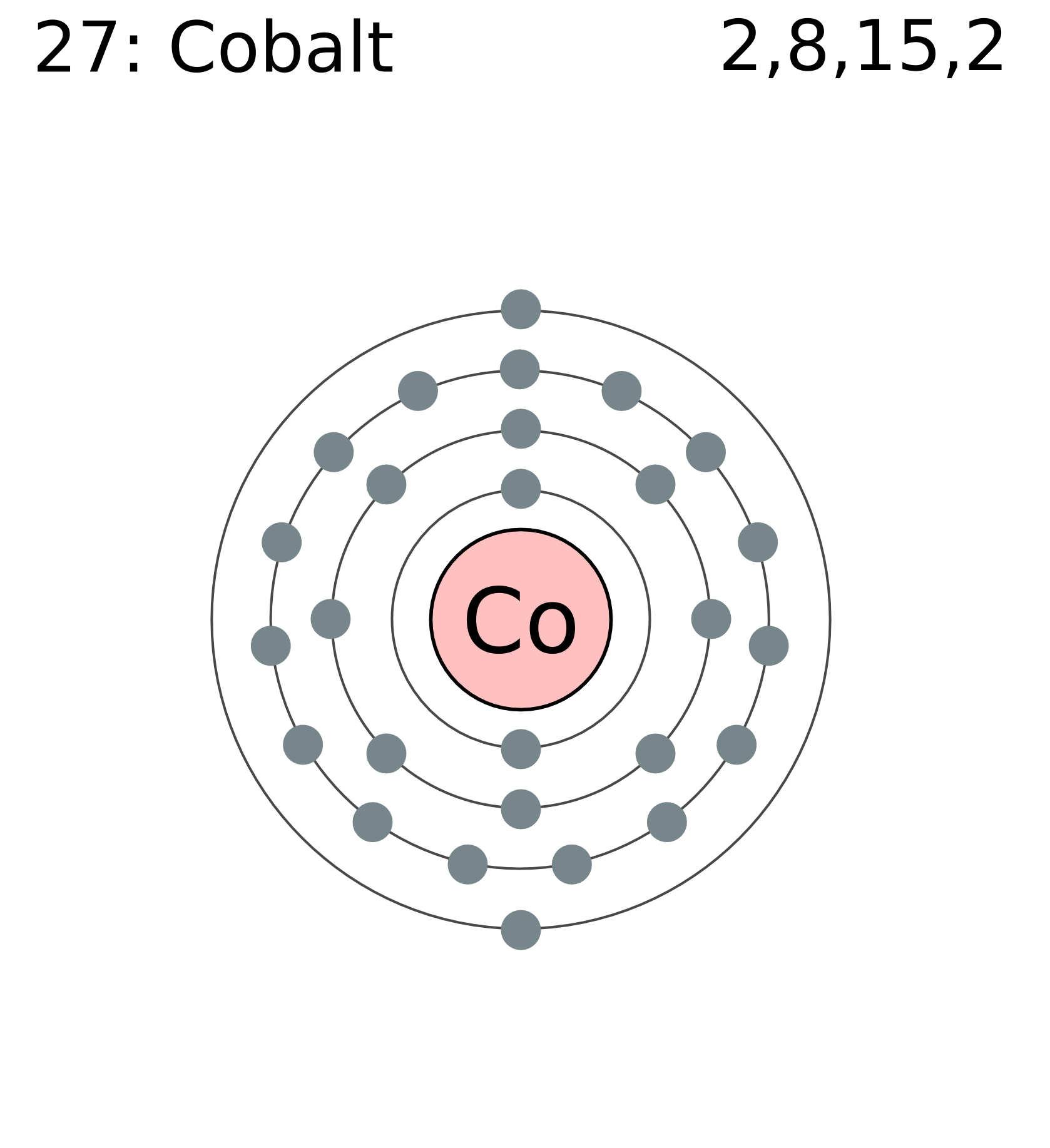
The electronic configuration of Cobalt will be 1s2 2s2 2p6 3s2 3p6 3d7 4s2.These effective nuclear charges, Z eff, are adapted from the following references: The electron configuration of Cobalt will end in d block as Cobalt is an inner transition metal. The electron configuration for Cobalt at ground state would simply be Co: Ar 4s 2 3d 7. How do you write the electron configuration for Cobalt? The s,p,d,f configuration for cobalt (Co) is 1s22s22p63s23p64s23d7, determined by the position of the element on the periodic table. The electronic configuration of Cobalt will be 1s2 2s2 2p6 3s2 3p6 3d7 4s2. What is the electronic configuration of Cobalt 27? What is the boiling Point of Cobalt in Kelvin?īoiling Point of Cobalt in Kelvin is 3200 K. 
Melting Point of Cobalt in Kelvin is 1768 K. What is the melting Point of Cobalt in Kelvin? What is the boiling Point of Cobalt?īoiling Point of Cobalt is 3200 K. Cobalt has 27 electrons out of which 4 valence electrons are present in the 3d7 4s2 outer orbitals of atom. Shorthand configuration Full configuration: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7 4s 2: Electron configuration The electronic configuration of cobalt is: 1s2 2s2. How many valence electrons does a Cobalt atom have?Ĭobalt has 4 valence electrons. What is the color of Cobalt?Ĭobalt is of Gray color. It is located in group 9 and period 4 in the modern periodic table. Cobalt is the 27 element on the periodic table. Therefore, the valence electrons of cobalt are nine. What is the position of Cobalt in the Periodic Table?Ĭobalt is a chemical element with the symbol Co and atomic number 27. This electron configuration shows that the last shell of cobalt has two electrons and the d-orbital has a total of seven electrons. Cobalt is a chemical element with symbol Co and atomic number 27. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. Cobalt (Co) 28: Nickel (Ni) 29: Copper (Cu) 30: Zinc (Zn) 31: Gallium (Ga) 32: Germanium (Ge) 33: Arsenic (As) 34. The abbreviated electronic configuration of Cobalt is 3d7 4s2. Find Abbreviated Electron Configuration For Cobalt related suppliers, manufacturers, products and specifications on GlobalSpec - a trusted source of. The condensed electron configuration is Ar. What is the abbreviated electronic configuration of Cobalt? The full electron configuration of a cobalt atom (Z 27) using s, p, d notation is 1s 2s 2p 3s 3p 4s 3d. Without referring to the periodic table, write the full electron configuration of the following elements. Cobalt is a chemical element with the symbol Co and atomic number 27. The element cobalt (Co) is in Period 4 and Group 9. The electronic configuration of Cobalt is 1s2 2s2 2p6 3s2 3p6 3d7 4s2. Electron configuration 3d 7 4s 2: Electrons per shell: 2, 8, 15, 2: Physical properties. For a Co 1+ ion coordinated to ligands with approximately octahedral symmetry, the 5 metal d-orbitals. What is the electronic configuration of Cobalt? Figure S6: Electronic configuration of the cobalt complex. Optical Properties of Cobalt Refractive IndexĪcoustic Properties of Cobalt Speed of SoundĬobalt Thermal Properties - Enthalpies and thermodynamics Refer to table below for the Electrical properties ofCobalt Electrical ConductivityĬobalt Heat and Conduction Properties Thermal Conductivity Hardness of Cobalt - Tests to Measure of Hardness of Element Mohs HardnessĬobalt is Conductor of electricity. Refer to below table for Cobalt Physical Properties DensityĨ.9 g/cm3(when liquid at m.p density is $7.75 g/cm3) In order to write the electron configuration we first need to know the number of electrons for the Cobalt (Co) atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
